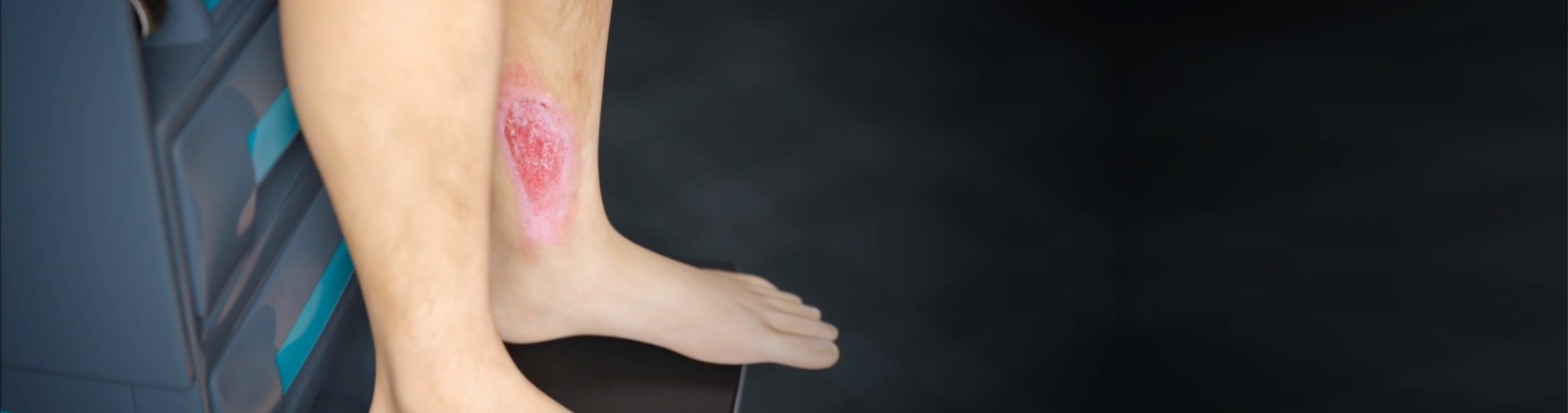
What Are Venous Leg Ulcers?
It’s important to get treatment for any unhealed wounds that form on the legs.
About Venous Leg Ulcers
Venous leg ulcers are a symptom of advanced vein disease. Certain factors put you at increased risk for venous leg ulcers, including varicose veins, obesity, standing for long periods of time (usually for work) and having a history of blood clots in your legs.
.png)
1 in 4 Americans 65+ will have a venous leg ulcer in their lifetime.1
What Causes Venous Leg Ulcers
Venous leg ulcers are caused by high pressure in the lower leg veins, called venous hypertension. When this pressure continues, it damages the skin, which breaks down and forms an ulcer. This video explains more about venous leg ulcers, why they form and what you can do about them.
Symptoms of Venous Leg Ulcers
Venous leg ulcers can be unsightly and painful wounds that develop around your ankle. They typically look like large, shallow wounds with uneven edges. Other symptoms2 can include:
.png)
References
- Margolis et al. J Am Acad Dermatol. 2002;46(3):381-386.
- https://health.clevelandclinic.org/leg-sores-wont-heal-whats-causing-your-venous-leg-ulcers/



.jpg)